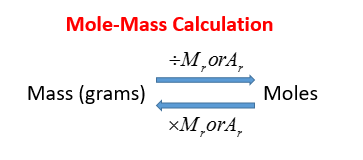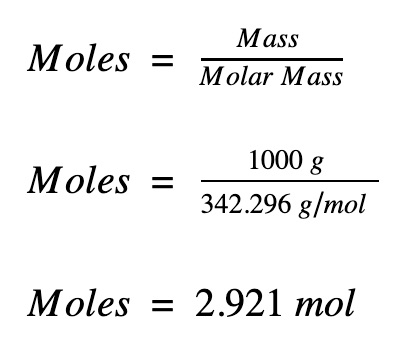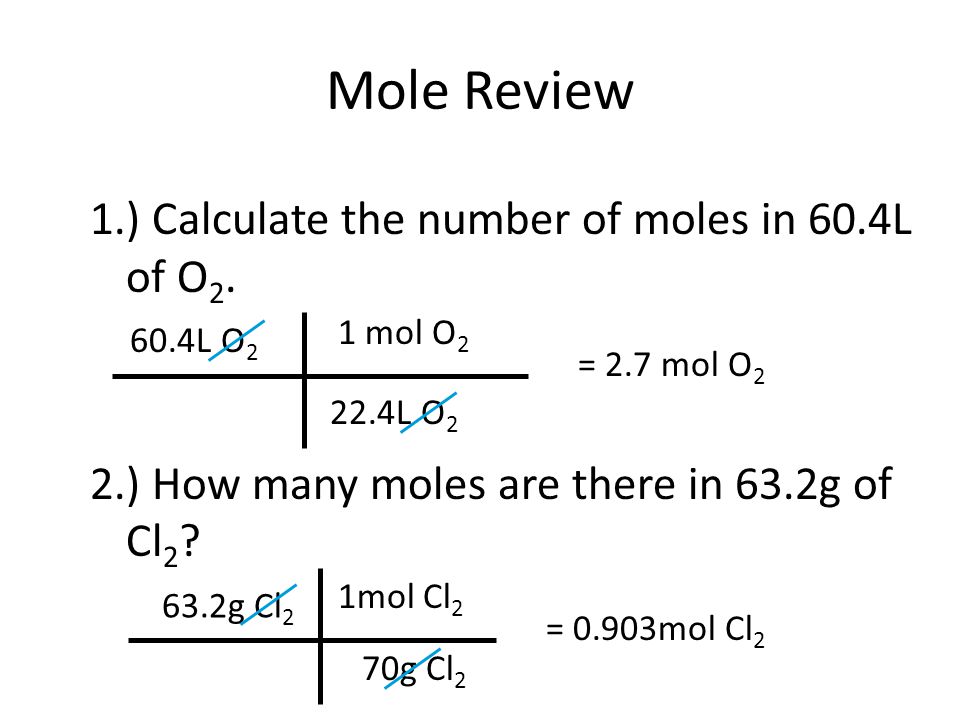
Mole Review 1.) Calculate the number of moles in 60.4L of O2. 2.) How many moles are there in 63.2g of Cl2? 1 mol O2 60.4L O2 = 2.7 mol O2 22.4L

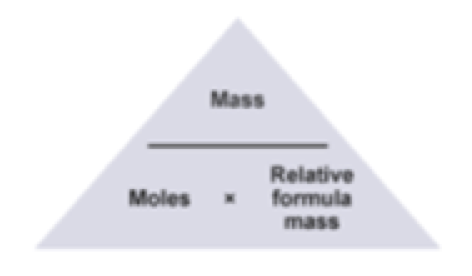
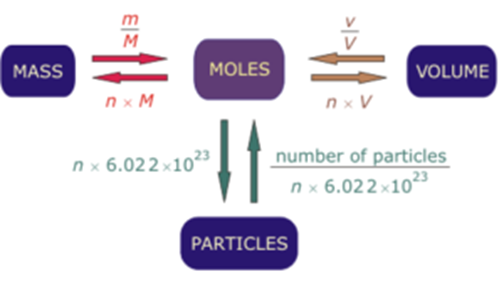
The Mole Formula Triangle Isolated On White Relationship Between Moles Mass And Molar Mass With Equations Stock Illustration - Download Image Now - iStock

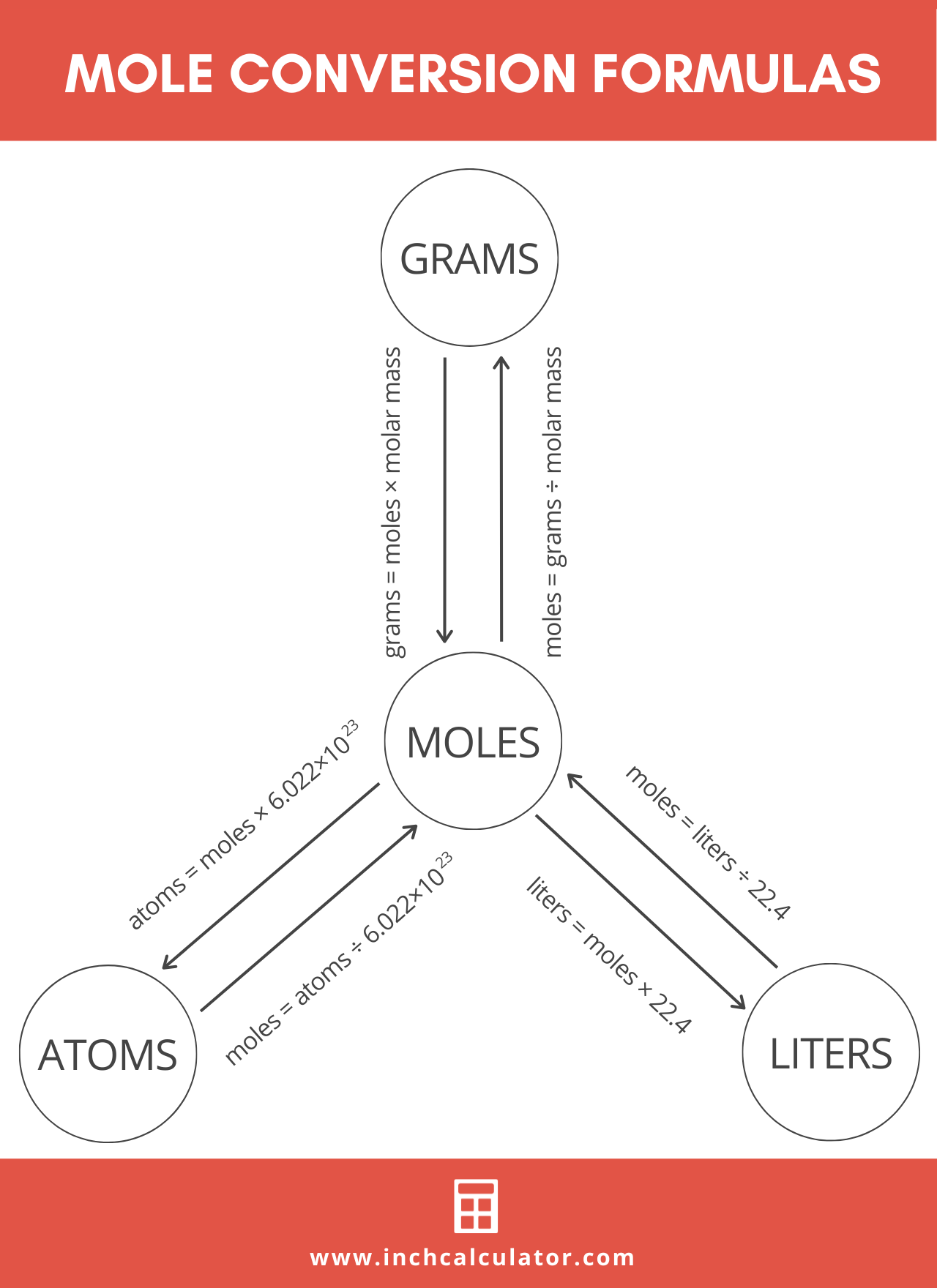
definition mole explained molar mass mol mols calculations how to read equations in moles deducing equations from reacting mole ratios questions gcse chemistry igcse KS4 science A level GCE AS A2 O

How to Find Moles of Product from Moles of Reactant using a Chemical Equation | Chemistry | Study.com