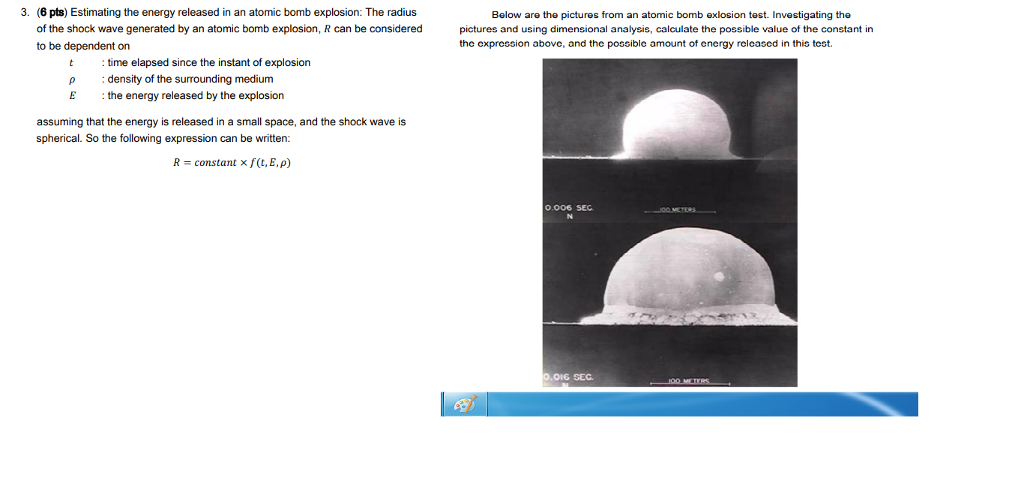
For complete combustion of ethanol, C2H5OH(l) + 3O2(g)→ 2CO2(g) + 3H2O(l) , the amount of heat produced as measured in bomb calorimeter is 1364.47 kJ/mol at 25^C . Assuming ideally, the enthalpy

SOLVED: The energy released through nuclear fission is typically much larger than typical chemical reaction Calculate the energy released for 1 mole of nuclear reaction that happens during an atomic bomb blast: