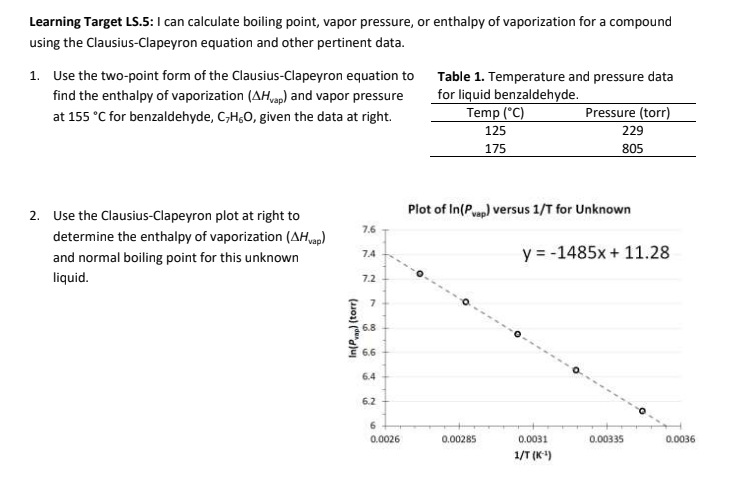
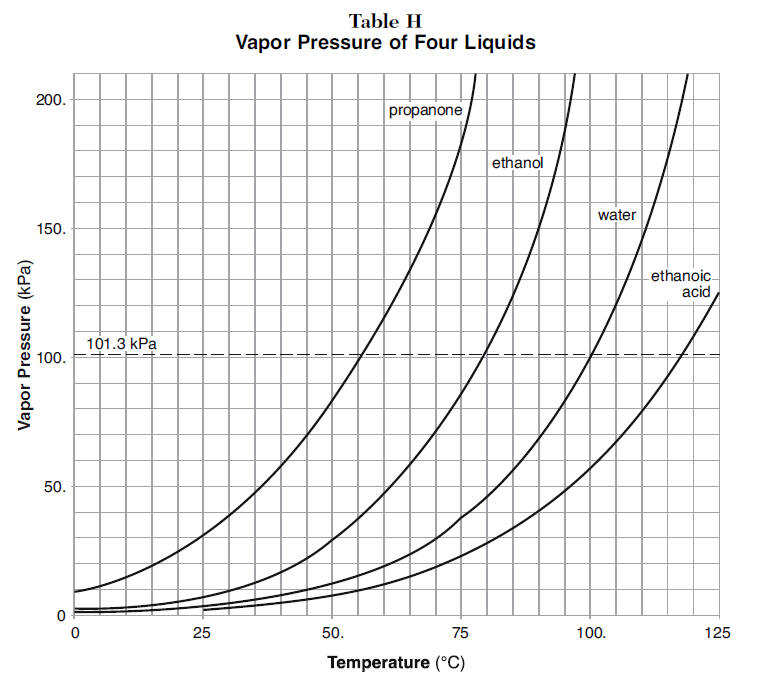
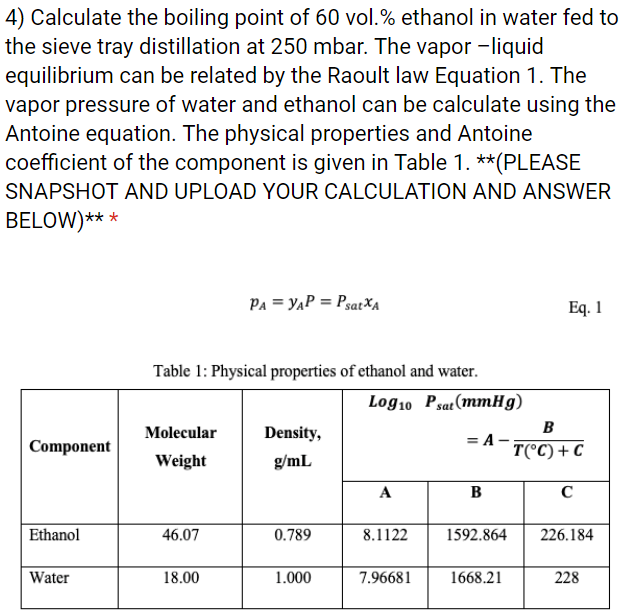
In a mountainous location, the boiling point of pure water is found to be 95 degrees Celsius. How many grams of sodium chloride much be added to 1 kg of water to

intermolecular forces - How can I determine the highest boiling point given a list of molecules? - Chemistry Stack Exchange

Determine the normal boiling point in K of a substance whose vapor pressure is 55.1 mmHg at 23.2^o C and has a ?H_vap of 32.1 kJ/mol. | Homework.Study.com

Boiling Point Elevation and Freezing Point depression - Example 2 ( Video ) | Chemistry | CK-12 Foundation
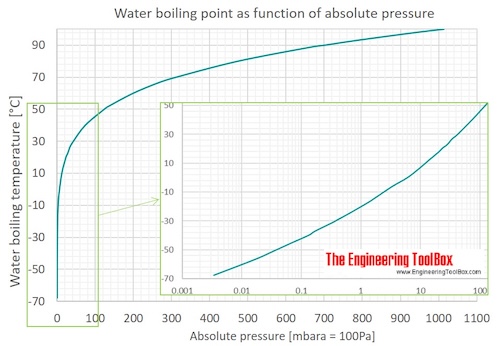
How does one calculate the boiling point of water at different pressures without a steam table? - Quora