Table 1 from In Situ Raman Spectroscopic Study of Gypsum (CaSO4·2H2O) and Epsomite (MgSO4·7H2O) Dehydration Utilizing an Ultrasonic Levitator. | Semantic Scholar
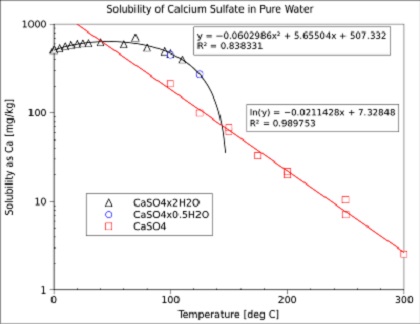
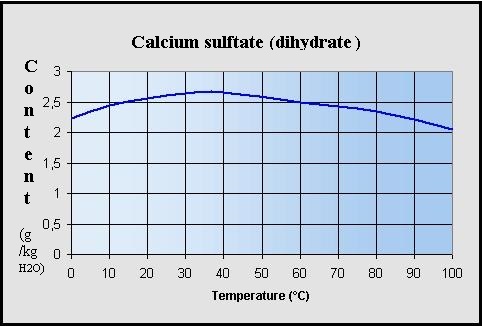
Comprehensive Thermodynamic Study of the Calcium Sulfate–Water Vapor System. Part 1: Experimental Measurements and Phase Equil
A crystallographic study of the low-temperature dehydration products of gypsum, CaSOa ' 2H2Oz hemihydrate CaSOr ' 0.50H2O, and 1
Membranes | Free Full-Text | Gypsum (CaSO4·2H2O) Scaling on Polybenzimidazole and Cellulose Acetate Hollow Fiber Membranes under Forward Osmosis

Experimental study of the dehydration reactions gypsum-bassanite and bassanite-anhydrite at high pressure: indication of anomalous behavior of H(2)O at high pressure in the temperature range of 50-300 degrees C. | Semantic Scholar

Dehydration Pathways of Gypsum and the Rehydration Mechanism of Soluble Anhydrite γ-CaSO4 | ACS Omega

Dehydration Pathways of Gypsum and the Rehydration Mechanism of Soluble Anhydrite γ-CaSO4 | ACS Omega
Investigations on dehydration and rehydration processes in the CaSO4 – H2O system at controlled time, temperature and humidity
A crystallographic study of the low-temperature dehydration products of gypsum, CaSOa ' 2H2Oz hemihydrate CaSOr ' 0.50H2O, and 1

Experimental Study of Hydration/Dehydration Behaviors of Metal Sulfates M2(SO4)3 (M = Sc, Yb, Y, Dy, Al, Ga, Fe, In) in Search of New Low-Temperature Thermochemical Heat Storage Materials | ACS Omega

Preparation and Application in HDPE of Nano-CaSO4 from Phosphogypsum | ACS Sustainable Chemistry & Engineering

Determination and Analysis of the Solubility of CaSO4·2H2O and α-CaSO4·0.5H2O in Formamide Aqueous Solutions at T = 303.15–363.15 K | Journal of Chemical & Engineering Data

In Situ Raman Spectroscopic Study of Gypsum (CaSO4·2H2O) and Epsomite (MgSO4·7H2O) Dehydration Utilizing an Ultrasonic Levitator | The Journal of Physical Chemistry Letters

Reaction control of CaSO4 during hydration/dehydration repetition for chemical heat pump system - ScienceDirect