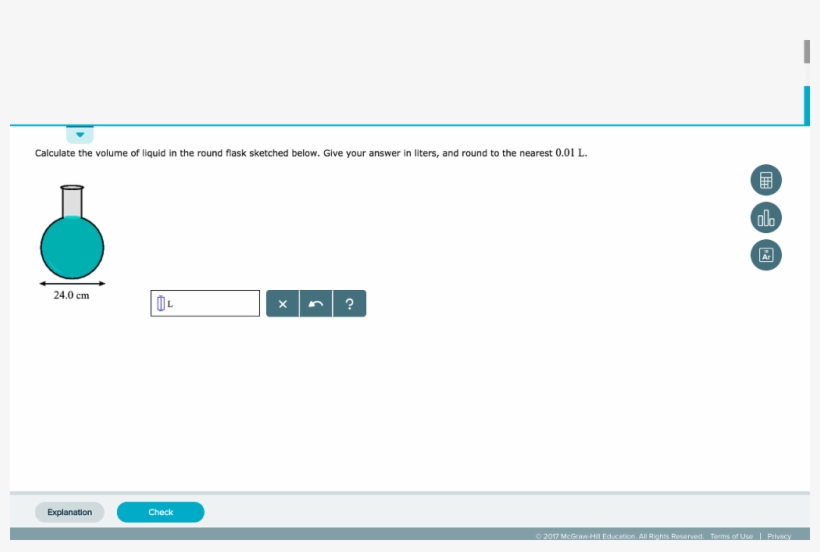
The volume of vapor in a flask can be determined by two methods: either by determining the mass of water required to just fill the flask completely or by using a graduated

SOLVED:To find the volume of a flask, the flask is evacuated so it contains no gas. Next, 4.4 g CO2 is introduced into the flask. On warming to 27^∘ C, the gas
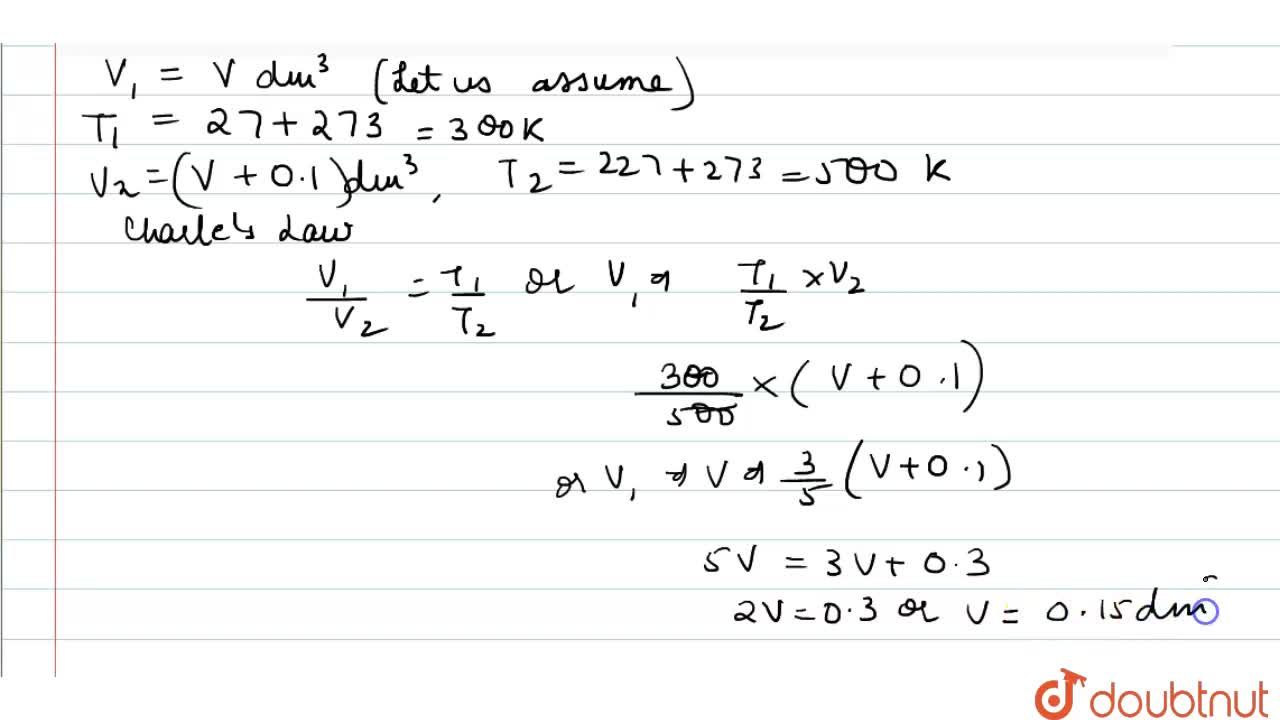
A flask was heated from 27^(@)C to 227^(@)C at constant pressure. Calculate the volume of the flask if 0.1 dm^(3) of air measured at 27^(@)C was expelled from the flask.

On , heating a flask at constant pressure, from 27^o C to 227^o C , 200mL of air was expelled out. what was the volume of the flask?

Two flask of equal volume are connected by a narrow tube (of negligible volume) are at `27^(@)C` and - YouTube

After completing this lesson you should be able to : Balanced equations show the mole ratio(s) of reactants and products. The molar volume is the same. - ppt download

After completing this lesson you should be able to : Balanced equations show the mole ratio(s) of reactants and products. The molar volume is the same. - ppt download

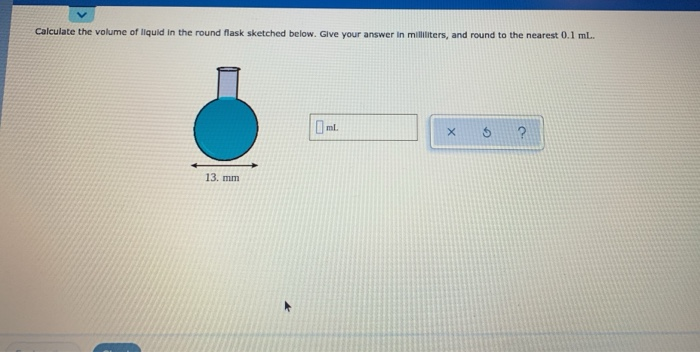
SOLVED:Finding the volume of a flask. A student obtained a clean dry glass-stoppered flask. She weighed the flask and stopper on an analytical balance and found the total mass to be 31.601




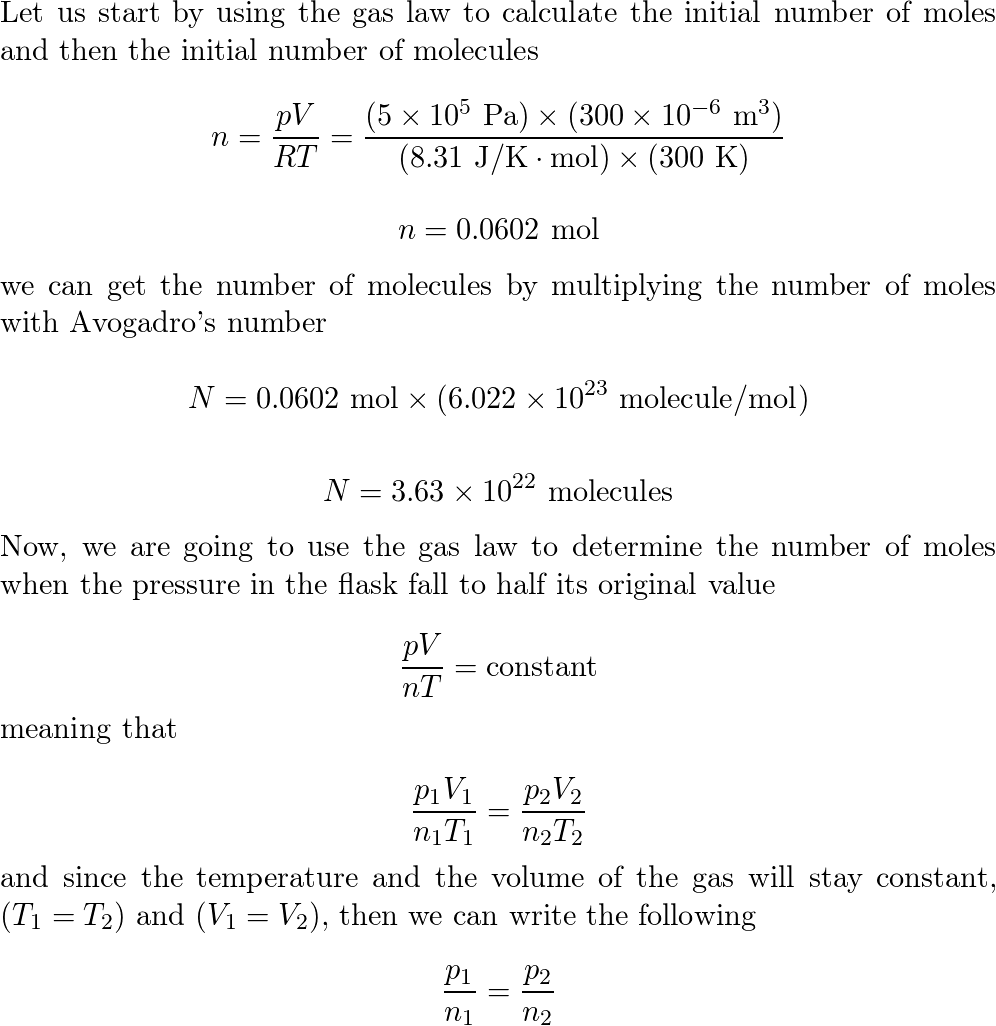
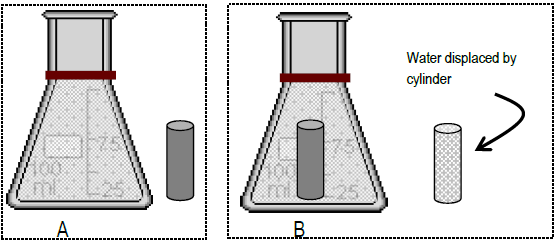


![ANSWERED] A flask with a sample of gas at room temp... - Physical Chemistry ANSWERED] A flask with a sample of gas at room temp... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/58199639-1659698160.4569945.jpeg)