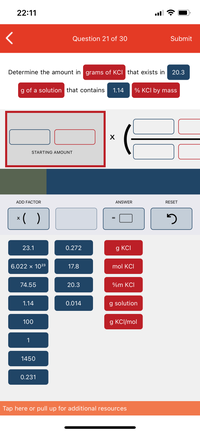
A solution of KCl has a density of 1.69 g mL^(-1) and is 67% by weight. Find the denisty of the solution if it is diluted so that the percentage by weight

Measurements of Vapor Pressures of Aqueous Solutions in the NaCl–KCl–H2O System from 493.15 to 693.25 K in a Fused Silica Capillary High-Pressure Optical Cell | Journal of Chemical & Engineering Data
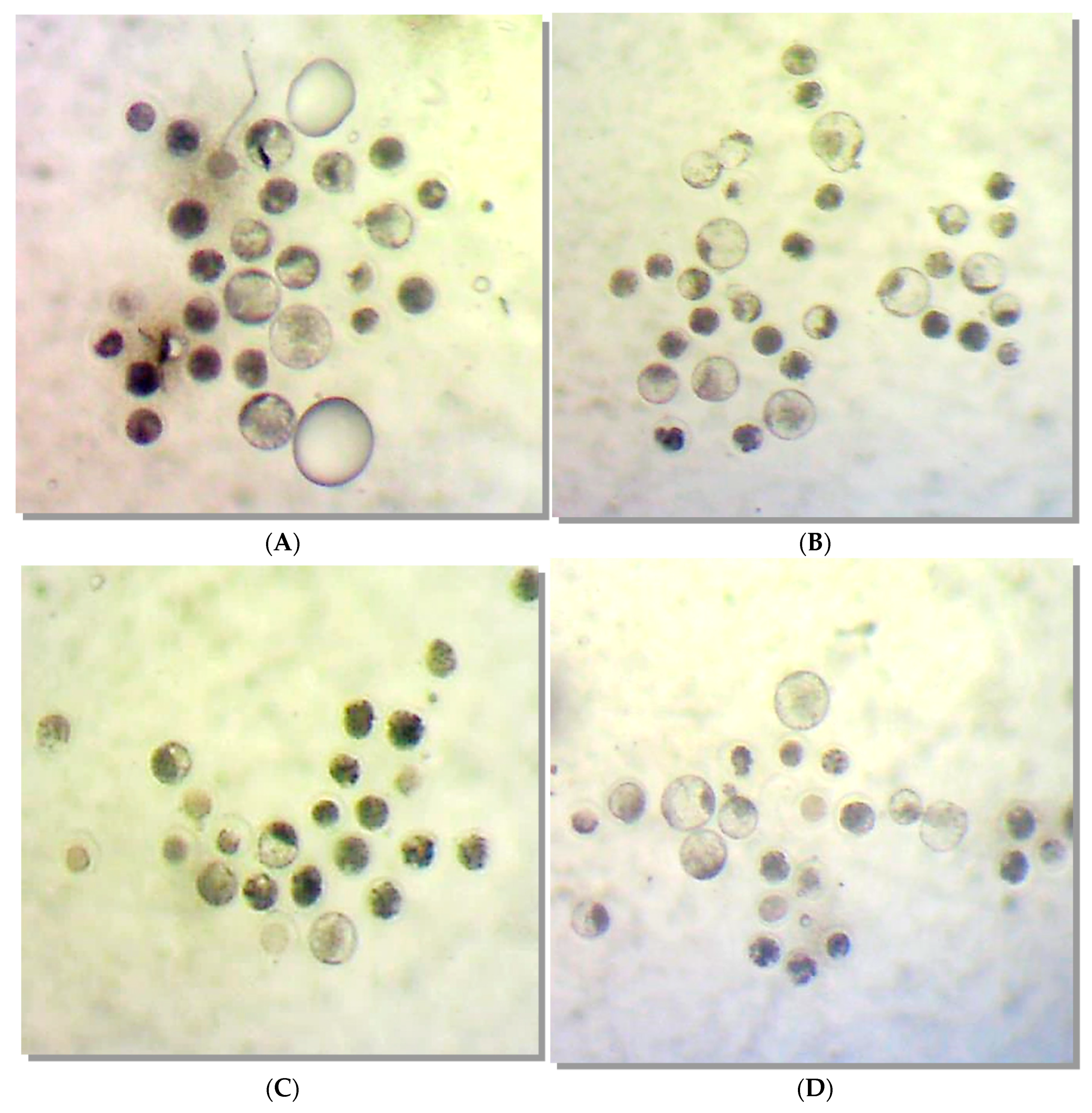
Molecules | Free Full-Text | Galectin-1 Used in Assisted Reproduction—Embryo Safety and Toxicology Studies
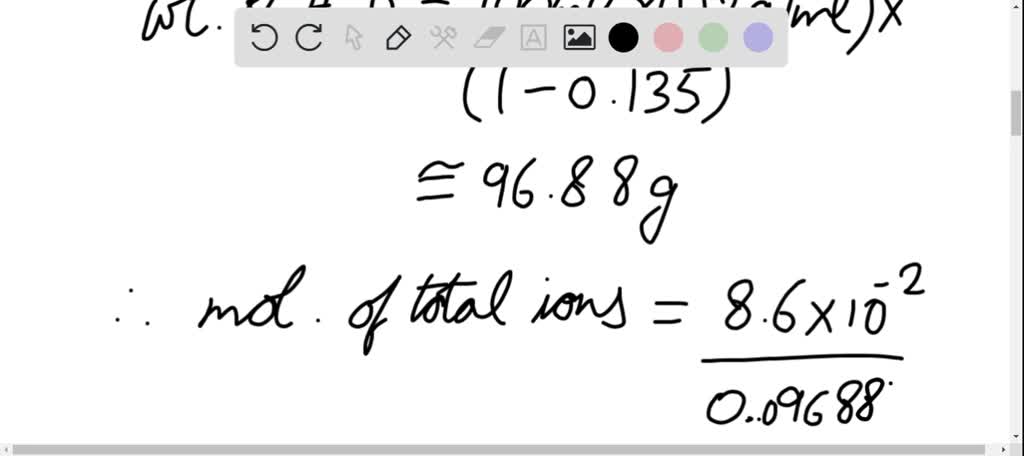
SOLVED:A 100.0 -mL aqueous sodium chloride solution is 13.5% NaCl by mass and has a density of 1.12 g / mL . What would you add (solute or solvent) and what mass

Ketamine decreases neuronally released glutamate via retrograde stimulation of presynaptic adenosine A1 receptors | Molecular Psychiatry
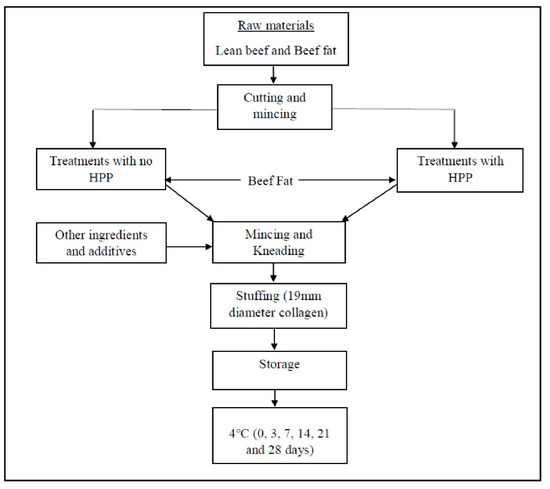
Processes | Free Full-Text | Effect of Partial Substitution of Sodium Chloride (NaCl) with Potassium Chloride (KCl) Coupled with High-Pressure Processing (HPP) on Physicochemical Properties and Volatile Compounds of Beef Sausage under

Quantification of Zeta-Potential and Electrokinetic Surface Charge Density for Colloidal Silica Nanoparticles Dependent on Type and Concentration of the Counterion: Probing the Outer Helmholtz Plane | The Journal of Physical Chemistry C

Utilization of Low-Concentration CO2 with Molecular Catalysts Assisted by CO2-Capturing Ability of Catalysts, Additives, or Reaction Media | Journal of the American Chemical Society
Change in the concentration of silicon-containing ions in the KCl melt... | Download Scientific Diagram

Calculate the molarity and molality of 20% aqueous ethanol (C2H5OH) solution by volume. (Density of solution = 0.96 g/mL)

A solution is prepared by dissolving 4 g of NaOH to give 500 ml of it. Calculate the molality of the solution.

The density of 3 M solution of NACl is `1.25 g mL^(-1)`. Calculate the molality of the solution. - YouTube

A solution of `KCl` has a density of `1.69 g mL^(-1)` and is 67% by weight. Find the denisty of ... - YouTube

Small molecule SWELL1 complex induction improves glycemic control and nonalcoholic fatty liver disease in murine Type 2 diabetes | Nature Communications

The density of a 10.0% by mass of KCl solution in water is 1.06 g/mL. Calculate molarity, molality and mole fraction of KCl in the solution.

Aq KCL #solution of #density 1.2 g/ml has a #molality of 3.30 mol/kg. find #molarity. #jeemains2021 - YouTube

The density of a 10.0% by mass of KCl solution in water 1.06 g/mL. Calculate molarity, molality and mole fraction of KCl in this solution respectively.

The density of a 10.0% by mass of KCl solution in water 1.06 g/mL. Calculate molarity, molality and mole fraction of KCl in this solution respectively.